 Finally, the Covishield vaccine has been approved for emergency use in India. Starting 2 January, a dry run commences for the vaccination program across the country. Hopefully, this will mark the beginning of the end for the century’s biggest threat to humanity. Use of COVID-19 sensors for temperature monitoring and control can help.
Finally, the Covishield vaccine has been approved for emergency use in India. Starting 2 January, a dry run commences for the vaccination program across the country. Hopefully, this will mark the beginning of the end for the century’s biggest threat to humanity. Use of COVID-19 sensors for temperature monitoring and control can help.
COVID-19 has indeed been the biggest economic disruption in a century. However, it is also turning out to be a giant digital-transformation catalyst for the healthcare sector, with a windfall for the logistics sector along the way.
The necessary thrust for this rapid transformation come from the urgency to overcome transportation challenges for some of COVID-19 vaccine candidates. Leading pharmaceutical companies like Pfizer and Moderna, along with their tier-1 logistics partners like UPS and FedEx, have been at the forefront of innovating processes and technologies for the purpose.
The key challenge, as we know, has been to maintain the vaccine vials at temperatures as low as minus 70 degree Celsius until they get thawed and administered to people.
Warehousing and logistics innovations fast-tracked
As we know, the need for storing and moving around truckloads (and airplane loads) of mass vaccine vials at –70 degree C posed a giant challenge at the very outset for pharma majors, particularly Pfizer. Clearly, the supply-chain team at Pfizer started to perfect a shipping solution soon as the scientists initiated the vaccine development program in the labs.
By October 2020, while positive breakthroughs were being achieved by scientists in vaccine development, Pfizer’s supply-chain team had worked out end-to-end logistics plans with partners such as United Parcel Service (UPS) and FedEx to deliver vaccine boxes to various parts of the USA as well as to different locations around the world.
Transportation and storage need two-pronged strategy
GPS-based temperature monitoring on the move was an obvious solution but putting it into practice was not an easy task.
As we know, special boxes were designed for transporting the vials. Each box was fitted with temperature sensors and a GPS device, which was used for tracking the temperatures inside the boxes from a monitoring center. Already, in instances where the temperature of a box crossed an upper (or lower) threshold, any further movement of the box was stopped and it was immediately recalled.
At pre-shipping stage, UPS, for example, had created an ultra-low temperature (ULT) freezer farm, from where the shipments would be done to the destined locations. These ULT freezers too were equipped with COVID-19 sensors for monitoring and maintenance purposes. As per an article posted by UPS on its website, “Cloud connectivity will enable sensor-based remote monitoring, control and predictive analytics across banks of ULT freezers. A continuous stream of data will detect performance characteristics to provide critical “onboard” information to ensure proper sample management, leading to increased efficacy of biologics.”
“Future ULT freezers will introduce guided access capabilities, which will allow the tracking and visualization of vials in laboratory management systems and at freezer access points,” the article, penned by Dusty Tenney, CEO of Stirling Ultracold, further noted. Stirling Ultracold is the company making the ULT freezers for the storage of vaccines.
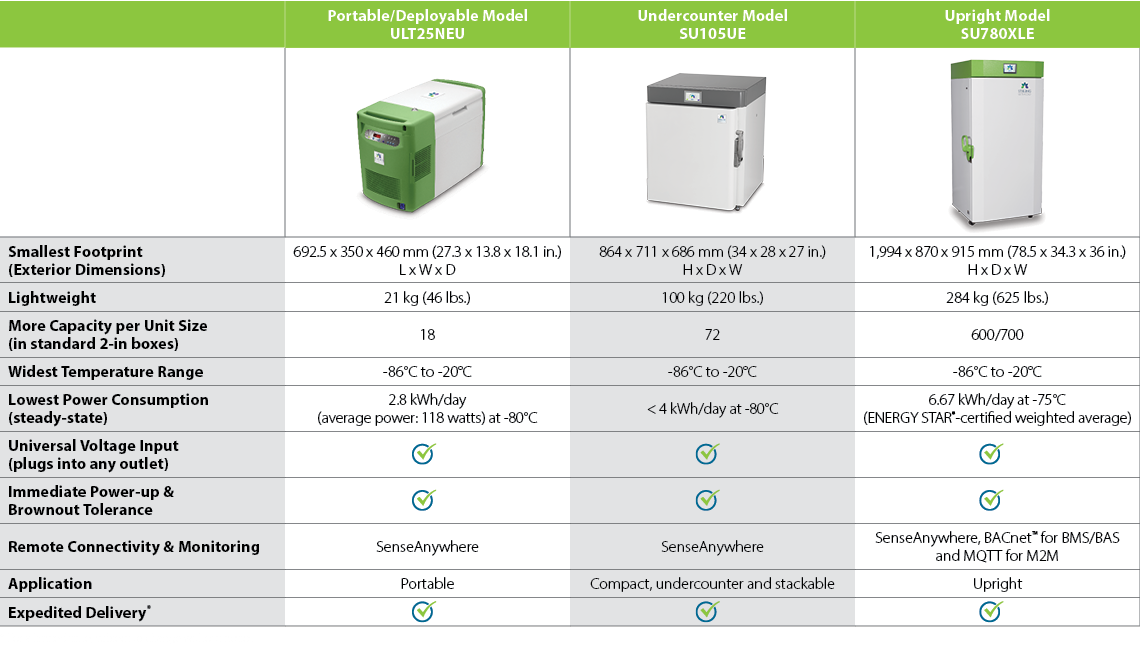
Figure source: Stirling Ultracold.
Global distribution may need to be ‘with the box’
The company has readied the ULT freezers in three different form factors (see Figure), each suited for the three key stages of a vaccine supply chain. The large ULT freezers are designed for manufacturing and warehousing bases for storing large number of vaccine vials; the mid-sized models are aimed at large pharmacies and hospitals; and the portable models are well-suited for serving the needs of local points of care, including remote and mobile points. All these models can maintain ultra-cold temperatures in the range of -80 degree C to -20 degree C, and are fitted with COVID-19 sensors for monitoring and control.
For a global distribution framework to be truly effective, all these different form factors need to be used at their respective target locations. In other words, while the big boxes will need to be deployed at warehouses, the mid-size boxes will need to installed at the hospitals in cities across the world ahead of the vial shipments. This important to ensure that vials can be stored at these hospitals when the shipments arrive there in ultra-cold boxes containing vials protected with dry ice packs. (Dry ice can only keep the boxes at ultra-cold temperatures of -70 degree C for up to five days, and is not readily and sufficiently available).
Likewise, the portable ULT models also need to be shipped in advance in satellite vaccination centers and remote locations.
All the time, remote monitoring of the ULT freezers and vial boxes is constantly being done to ensure that the efficacy of the vaccines is not lost due to change in box or freezer temperature before the vial being thawed for use.
While vanilla GPS solutions are being used for now, there is enough room for involving technologies like robotics and internet of things (IoT) in the delivery and monitoring process.
For example, if we consider the process of changing the dry ice in boxes, a human intervention could be time-consuming and hence cause the temperature of vials to rise to detrimental levels. However, with the use of robotics, this task can be accomplished in a safe and assured manner. As far as IoT is concerned, building management system (BMS) IOT connectivity is already available for Stirling’s ULT freezers.
The ULT freezers as well as shipment boxes for vaccine vials come fitted with COVID-19 sensors that can have battery lifespans of up to 10 years and support datalogging in the cloud, which makes them suited for real-time remote monitoring. For example, in case of Pfizer’s COVID-19 vaccine candidate, the solution comprises SenseAnywhere’s AiroSensors and Pt100 smart probes.
Why other vaccines too need digital monitoring and upkeep
Covishield, the vaccine candidate from Pune-based Serum Institute of India, may not require a very stringent temperature control and monitoring mechanism. The vaccines, which received the emergency-use approval and for which the dry run commences on 2 January 2021, can be safely stored in regular refrigerators. The storage temperature requirement for Covishield is 2–8 degree C, which also gives it a huge edge over other vaccine candidates in India.
Nevertheless, use of COVID-19 sensors can doubly ensure that the vials are constantly being maintained at the required temperature range. This is because power supplies can be quite unpredictable in smaller cities and towns and not all pharmacies and refrigerators are supported with power-backup facilities. By mandating that refrigerators used for storing vials be retrofitted with COVID-19 sensors for monitoring the temperatures, governments and health watchdogs can determine the efficacy efficiency of a vaccination program. In case of power failures and ineffectiveness of refrigerators, the necessary corrective actions may be taken in a timely manner.
Warehousing and distribution companies engaged in the logistics of COVID-19 vaccines would particularly need to get equipped with appropriate sensor mechanisms. It goes without saying that IT teams at these organizations will have a special role to play in ensuring integrated and foolproof solutions in this context.
See also How smartphones could be Covid-19 testing game changers











0 Comments